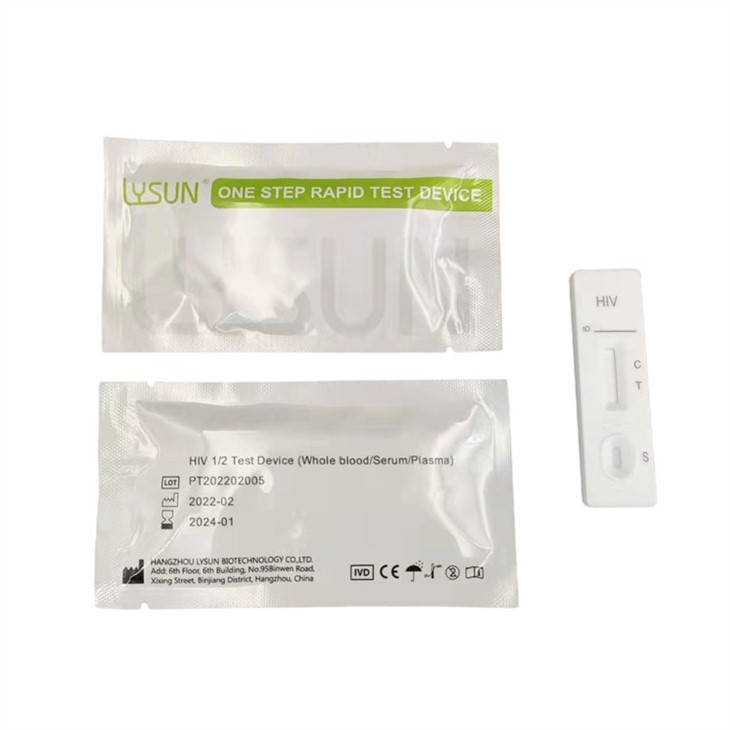
|
INTENDED USE |
HIV 1 2 O Rapid Test (Whole Blood/Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of antibodies to HIV 1 and/or HIV 2 in whole blood, serum or plasma.
|
SUMMARY |
HIV is the etiologic agent of Acquired Immune Deficiency Syndrome (AIDS). The virion is surrounded by a lipid envelope that is derived from host cell membrane. Several viral glycoproteins are on the envelope. Each virus contains two copies of positive‐sense genomic RNAs.
|
PRINCIPLE |
HIV 1 2 O Rapid Test (Whole Blood/Serum/Plasma) is a qualitative, membrane based immunoassay for the detection of antibodies to HIV 1/2 in whole blood, serum or plasma. The membrane is pre‐coated with recombinant HIV antigens. During testing, the whole blood, serum or plasma specimen reacts with HIV antigen coated particles in the test strip. The mixture then migrates upward on the membrane chromatographically by capillary action and reacts with recombinant HIV antigen on the membrane in the test line region. If the specimen contains antibodies to HIV 1 and/or HIV 2, a colored line will appear in the test line region, indicating a positive result. If the specimen does not contain HIV 1 and/or HIV 2 antibodies, a colored line will not appear in the test line region, indicating a negative result. To serve as a procedural control, a colored line will always appear in the control line region, indicating that proper volume of specimen has been added and membrane wicking has occurred.
|
How to use |

|
PRECAUTIONS |
*For professional in vitro diagnostic use only. Do not use after expiration date.
*Do not eat, drink or smoke in the area where the specimens or kits are handled.
*Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout testing and follow the standard procedures for proper disposal of specimens.
*Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are being tested.
*Humidity and temperature can adversely affect results.
|
Packing Details |
|||
|
|
HIV 1 2 O Rapid Test |
HIV-W101 |
Test strips |
|
|
Buffer |
||
|
|
Test cards |
||
|
|
Disposable specimen droppers |
||
|
|
Package insert |
||
|
Delivery |

|
Company Profile |
Hangzhou Lysun Biotechnology Co., Ltd.
Hangzhou Lysun Biotechnology Co., Ltd. is a manufacturer of modern medical equipment and reagents with a reliable treatment system. Overall, Lysun Biotechnology is committed to improving the quality and design of laboratories and hospitals.
Lysun Biotechnology focuses on development, manufacturing, purchasing and working with a strong R&D team. Since its establishment in 2018, LYSUN has obtained 8 national software rights, 8 design patents, 1 available patent status, and 4 image patents.
Lysun Biotechnology will continue to focus on the in vitro signal industry and promise to be a leading manufacturer of medical diagnostic equipment.
|
Services |
- 7-24 Internet connection;
- Laboratory tests and site quality control.
- Registration service.
- OEM is popular.
- CE and ISO13485 certification;
- High quality products and fast shipping.
- The technical R&D team has after -sales service.